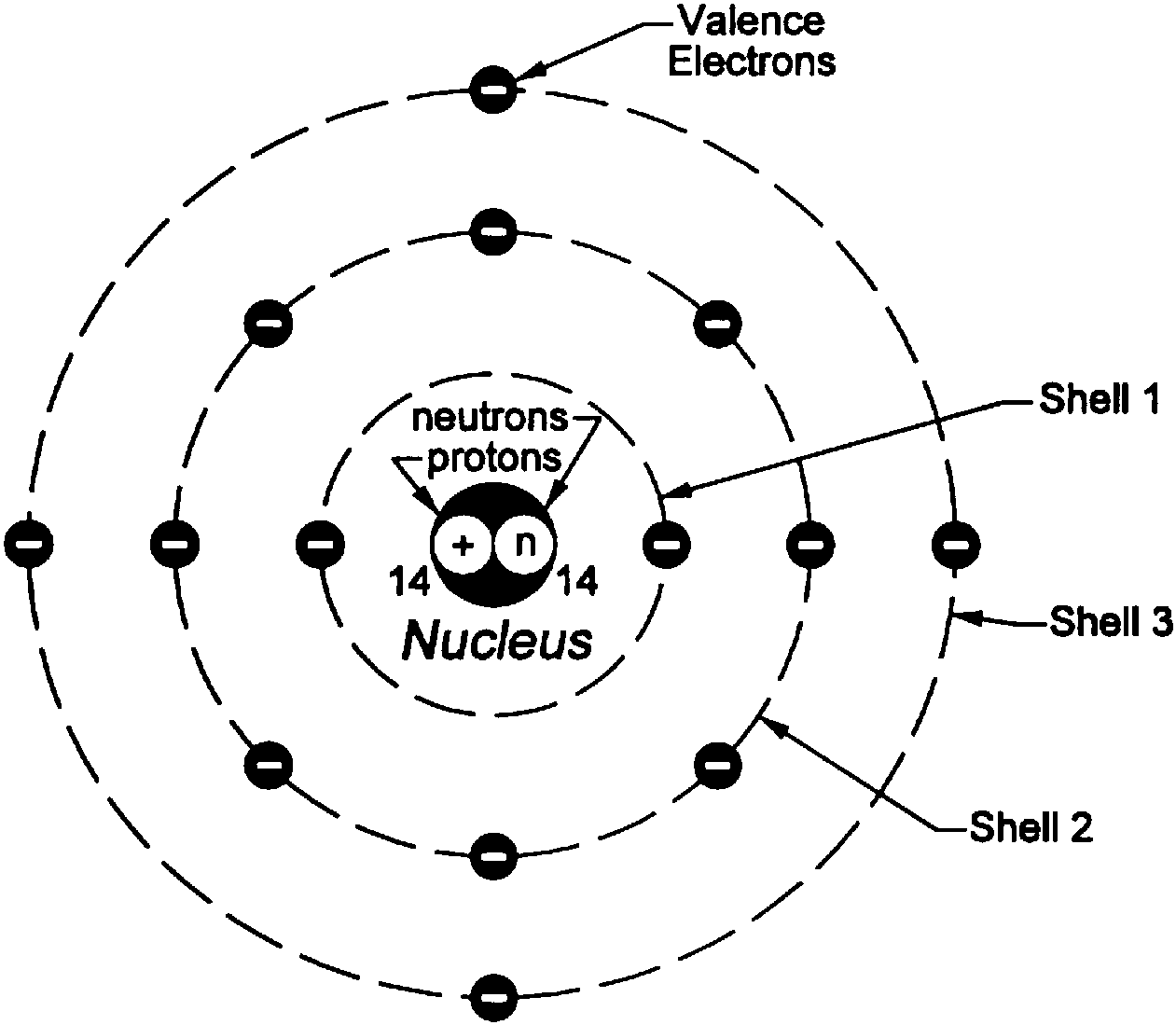
  Rutherford suggested that perhaps the atomic system was analogous to our Solar System, where the electrons revolve around the nucleus like planets revolving around the Sun. The volume of empty space between an atom’s electrons and its nucleus is so huge that if the atom were expanded to the size of a baseball stadium, its nucleus would be the size of a baseball. Rutherford realized that most of the atom was filled with empty space, but at the center was a dense, point-like concentration of positive charge. This view held true until Ernest Rutherford showed that when positive particles are shot at an atom, most of them pass straight through, but a few are observed to be deflected at a large angle. The model is popularly known as the plum or raisin pudding model. He suggested that the negative particles, which he called electrons, were like seeds embedded in a positively charged watermelon. Thomson devised what became the very first model of an atom. He immediately realized that to account for matter’s stability, there must be a net positive charge to neutralize the negativity. If atoms were merely a cluster of negative charges, then chairs, tables, you and I would be anything but stable. However, the subatomic particle he discovered was negatively charged. Thomson who first discovered that the atom wasn’t indivisible after all, a notion believed to be true for centuries. What extraordinary feat did Niels Bohr accomplish to deserve this prestigious honor, and well, a Nobel Prize? BOHR ATOM MODEL FREEBohr was rewarded with a lifetime supply of free beer that would pour out of a tap at his whim. The house was connected to the brewery by a pipeline. For his exemplary contributions to science, the Carlsberg brewing company decided to give him a house situated right next to one of their breweries. Niels Bohr was a Danish physicist and is considered one of the founding fathers of quantum mechanics, precisely old quantum mechanics. The difference between the energies of those orbits would be equal to the energy of the photon. 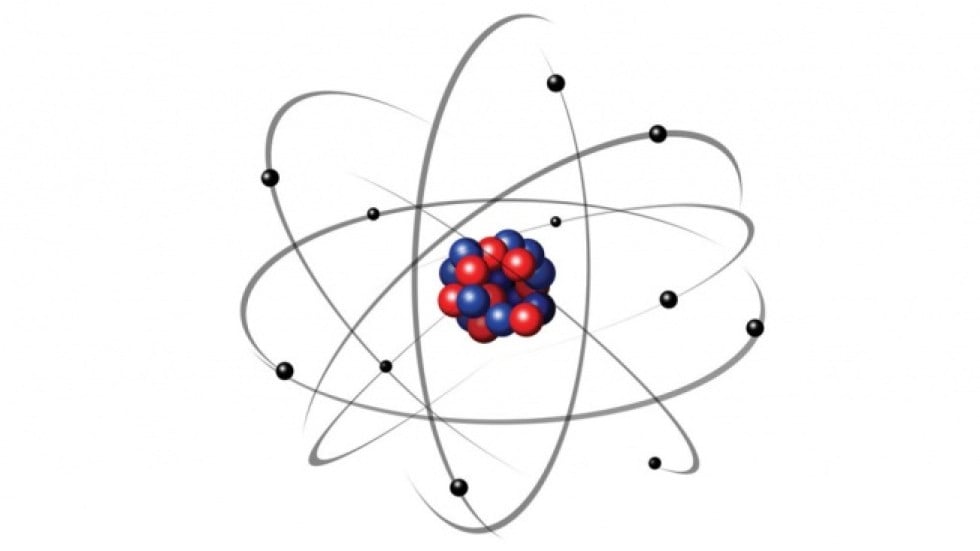
The atom would radiate a photon when an excited electron would jump down from a higher orbit to a lower orbit. Niel Bohr’s Atomic Theory states that – an atom is like a planetary model where electrons were situated in discretely energized orbits. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
